A white paper describing how IHE’s Methodology contributes to the broad adoption of HL7®FHIR® in support of the EHDS
Publications


IHE-Europe 20th anniversary
About 20 years ago a small group of like-minded Health IT experts in Europe, who believed that following the concepts of IHE would help leverage the benefits of information technology for healthcare, came together to found IHE-Europe.

mHealth: Joint recommendations for an effective deployment.
COCIR and IHE-Europe have worked closely with other key stakeholders to recommend the inclusion of use cases to demonstrate how utilising IHE Profiles improves the interoperability of mHealth solutions.

Imaging Taskforce Survey: Initial Results on Image Sharing Platforms
IHE-Europe European Affairs Committee recently undertook an Imaging Taskforce Survey to better understand how IHE Profiles are implemented in image sharing platforms throughout Europe.

Initial Contribution on European Commission recommendation
IHE drafts a white paper developing a vision for interoperability that answers the European recommendation for developing a “common approach in support of mobile applications to help address the COVID-19 crisis”.

IHE Perspective on the European Union GDPR
This Whitepaper shows how IHE helps understand the General Data Protection Regulations - GDPR - the influence it has on the use of IHE Profiles and the actions that need to be undertaken as a priority to comply and stay compliant.

Whitepaper on Connectathon - new edition 2020
What is the IHE Connectathon? The objective of the IHE Connectathon is to test participating systems’ conformity to IHE Profiles by using validators and the Interoperability between systems, following clinical workflows.
IHE Profiles enable seamless and secure exchange of health information for both providers and patients across the globe, and provide access to a world-wide eHealth market.

IHE-Europe Flyer
IHE is a world-wide initiative that allows Healthcare IT system users and suppliers to work together to enable interoperability of IT systems.
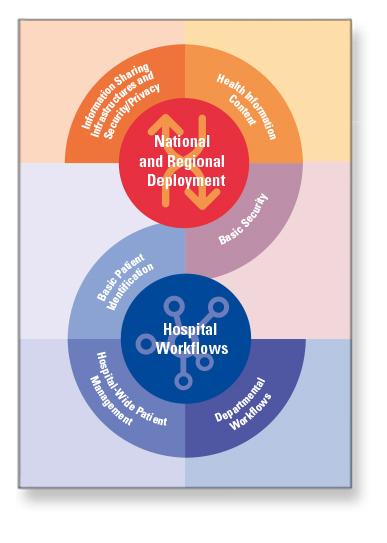
27 Profiles Flyer
The European Commission has acted to reinforce quality patient care and facilitate access to medical information by healthcare professionals. As part of the procurement process it has endorsed the application of reliable means of electronic exchange of information using recognised profiles and standards – 27 IHE Profiles.

Gazelle Flyer
The Gazelle Test Platform is at the heart of conformity and Interoperability testing.

Conformity Assessment flyer for Vendors
IHE Conformity Assessment is ideal for vendors who are in search of more rigor and who passed the IHE Connectathon tests within the prior two years. The product must be either market released or planned to be released within six months.

Conformity Assessment flyer for Users
IHE Conformity Assessment is ideal for the acquistion of new products or upgrades to existing products with requirement to comply with IHE Profiles. Conformity Assessment testing is based on an ISO/IEC 17025 quality system in accordance with IHE International Conformity Assessment.

IHE's 2020 Vision
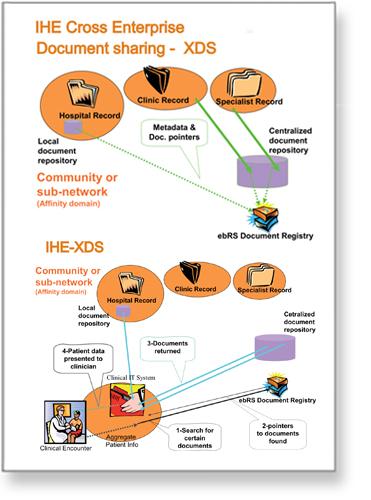
Fact sheet on XDS
Sharing clinical documents using IHE specifications
IHE Cross-Enterprise Document Sharing (XDS) is an international solution defined by the Integrating the Healthcare Enterprise (IHE) of using an ebRegistry (OASIS and ISO standard) to share electronic documents.



